A study of the biological basis for jetlag has revealed a single gene that controls our body clock. The discoverers hope this will lead to drugs that will make the lives of long distance travelers more comfortable, but could also make those of shift-workers longer.
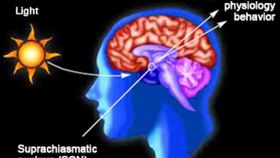
The rest of this article is behind a paywall. Please sign in or subscribe to access the full content.The cells in our body experience a rise and fall in protein levels over a 24 hour period, setting us up to sleep at roughly the same time each day. This cycle is driven by the suprachiasmatic nucleus (SCN) a part of the brain notable for the density of its 20,000 neurons and the frequency of communication between them.
Spending time in sunlight after an east-west flight is only partly successful in resetting your body clock because less than half the neurons in the SCN respond to light exposure and it takes time for these to bring the other cells round. The stability of the SCN, beneficial when our body clocks only needed to be reset seasonally, has become problematic in a faster paced age.
When researchers at the Salk Institute for Biological Studies disrupted the light-dark cycles mice were exposed to they observed changes in the expression of thousands of genes, 213 unique to the SCN. Of these 13 were masterswitches, they report in eLIFE, coding for molecules that control other genes.
Of these one, Lhx1, stood out, being suppressed when the mice were exposed to light. Lhx1's role in brain development makes it essential to life, but lead author Shubhroz Gill said, "No one had ever imagined that Lhx1 might be so intricately involved in SCN function.” It turns out that part of Lhx1's role is to make neurons communicate, without it the vital interplay between neurons breaks down.
While an absence of Lhx1 is fatal, a reduced amount can have its benefits. Mice with lower Lhx1 expression responded more quickly to changes in lighting hours that model major timezone shifts. It seems Lhx1 provides inertia, by keeping SCN neurons in step and therefore more resistant to change. Suppressing Lhx1 long-term would be damaging, as “behavior rhythms gradually deteriorate under constant condition,” according to the paper. However, a drug whose effects wear off quickly would be in high demand.
While jetlag may be the most widely experience effect of too much Lhx1, the consequences of shift work are far more problematic, with serious and often life-threatening health damage to the millions of workers forced to change their hours regularly. Disorders of the SCN are also one cause of insomnia.
Reduction in Lhx1 also reduced the activity in many other genes, including some where no connection had previously been suspected. One of these, Vip, is able to compensate for a loss of Lhx1. Vip is a very important protein for the researchers because, being released outside cells, rather than inside like Lhx1, it is easier to target.
"If we find a drug that will block the Vip receptor or somehow break down Vip, then maybe that will help us reset the clock much faster," says team leader Dr Satchidananda Panda.