First intestines, now stomachs! Last week, researchers reported growing miniature human small intestines in mice. Well, this week, another team out of Cincinnati say they’ve grown 3D stomach tissue in the lab out of human stem cells. The work was published in Nature this week.
The rest of this article is behind a paywall. Please sign in or subscribe to access the full content.Ulcers, stomach cancer, and other gastrointestinal diseases affect 10 percent of the world’s population, and their development is linked to chronic Helicobacter pylori infection. But because of the differences between people and lab animals such as mice and flies, existing live models aren’t ideal for studying human stomach development and disease.
To create a more realistic model, a team led by University of Cincinnati’s Yana Zavros and James Wells from Cincinnati Children’s Hospital Medical Center started with human stem cells -- specifically pluripotent stem cells, which have the potential to become many different kinds of cells in the body. After that, the key was to identify the steps involved in normal stomach formation during our embryonic development.
“Until this study, no one had generated gastric cells from human pluripotent stem cells (hPSCs),” Wells explains in a news release. “In addition, we discovered how to promote formation of three-dimensional gastric tissue with complex architecture and cellular composition.”
By guiding the stem cells through all these natural, sequential processes in a petri dish, the team coaxed the hPSCs toward becoming stomach tissue. The researchers focused on a pathway of interactions that acts as a switch between growing tissues in the intestine and in the antrum (the stomach’s outlet to the small intestine), Nature News reports. When the cells were three days old, they added proteins to suppress this pathway.
Over the course of a month, these steps resulted in the formation of so-called human gastric organoids. These miniature stomachs are only about three millimeters in diameter, and their 3D structure contains different kinds of cell types with functional characteristics resembling those seen in our stomachs.
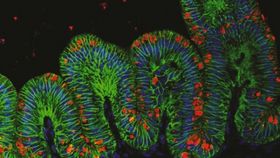
 Pictured to the right, an immunofluorecent image of human gastric organoid (green) and H. pylori (red).
Pictured to the right, an immunofluorecent image of human gastric organoid (green) and H. pylori (red).
Then, to demonstrate the capability of these stem-cell-derived stomach tissues to act as a model for studying diseases, the team unleashed the bacteria. Given the chance, H. pylori infected the tissue rapidly: Within 24 hours, the bacteria had triggered biochemical changes to the organ. The gastric organoids faithfully mimicked the early stages of gastric disease, which includes the activation of a cancer gene called c-Met and the rapid spread of infection in epithelial tissues.
The team hope their approach can help identify what drives normal stomach formation in people in order to understand what goes wrong when the stomach doesn’t form the right way. The new system can also improve gastric-bypass procedures and help with drug development for conditions ranging from cancer to diabetes. Maybe one day, the work could lead to replacement stomachs.
Images: Kyle McCracken