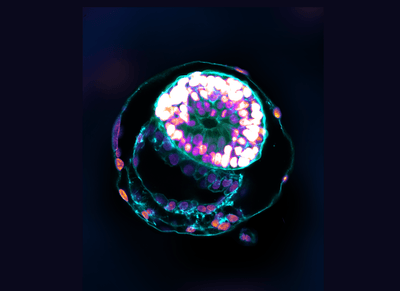
Scientists have created "complete" human embryos using stem cells in a lab and managed to grow them outside the womb for 14 days. Fear not, this is not an attempt to make a living Franken-baby in a test tube or anything of the kind. Instead, the researchers aim to provide a model that can help increase our understanding of a crucial moment in embryo development that impacts the risk of miscarriage and birth defects.
The rest of this article is behind a paywall. Please sign in or subscribe to access the full content.Two teams of scientists – one from Yale School of Medicine and another from the University of Cambridge – have recently announced success stories in creating synthetic embryo-like models, but this latest research from the Weizmann Institute of Science in Israel goes a step further as their embryo models have a significantly more complex, self-organized structure.
The work aims to shed light on a stage of embryo development called gastrulation that occurs two to three weeks after conception. It’s a critical stage of development, but scientists know very little about this fundamental turning point in early development as the microscopic embryo is buried in the womb at this time.
“The drama is in the first month, the remaining eight months of pregnancy are mainly lots of growth,” Professor Jacob Hanna at the Weizmann Institute of Science, who led to the project, said in a statement sent to IFLScience.
“But that first month is still largely a black box. Our stem cell–derived human embryo model offers an ethical and accessible way of peering into this box. It closely mimics the development of a real human embryo, particularly the emergence of its exquisitely fine architecture,” Hanna said.

Just like previous studies, the embryo models were not created using sperm, fertilized eggs, or a uterus. The researchers managed to coax human stem cells into organizing themselves into synthetic embryo-like models that closely resemble a human embryo.
Based on their previous work with mouse embryos, the team took pluripotent stem cells – "blank slate" cells that have the potential to differentiate into many different cell types – and made them revert these cells to an earlier "naive" state.
Through a series of intricate steps, this enabled them to produce self-assembling models that closely mimicked the 3D architecture and key developmental landmarks of human embryos from 7 days after fertilization, around the time it implants itself in the womb, to 14 days after fertilization.
“An embryo is self-driven by definition; we don’t need to tell it what to do – we must only unleash its internally encoded potential,” explained Professor Hanna.
“It’s critical to mix in the right kinds of cells at the beginning, which can only be derived from naïve stem cells that have no developmental restrictions. Once you do that, the embryo-like model itself says, ‘Go!’”
The researchers claim their work goes further than the previous attempts as the model human embryos contain key cell types that are essential to the embryo’s development, such as those that form the placenta and the chorionic sac, which the other studies did not.
Other experts in the field have commented on how this added step could significantly boost the potential of embryo-like models in scientific research – but it might also raise further ethical dilemmas owing to their complexity.
“From my point of view, the issue to emphasize is that the system reproduces the natural situation remarkably well and, unlike all the other reports that have appeared during the summer, this is a landmark study that opens up new avenues for research into human development and the study several aspects of fertility and disease,” Professor Alfonso Martinez Arias, ICREA Senior Research Professor at Pompeu Fabra University in Spain, explained to the Science Media Centre.
He points out that previous attempts were merely “mangled groups of cells” rather than anything resembling the structures we see in living beings.
“I expect the work to raise ethical issues but, unlike earlier claims, this time with a real basis to think about the questions that emerge,” he added.
The new study is published in the journal Nature.