Researchers have raised the possibility of a new form of ice, one that would break the record set two years ago for low-density solid water.
The rest of this article is behind a paywall. Please sign in or subscribe to access the full content.Anyone with access to refrigeration is familiar with one form of frozen water, and walking outside in winter at high latitudes or altitudes introduces us to another. We might expect the possible ways to turn dihydrogen monoxide into a solid ends there, but the flexibility of the hydrogen bonds in water have allowed the creation of 17 crystalline phases of water. Now researchers think they have designed another.
Two years ago a French-German collaboration produced ice XVI, the least dense form of ice known. However, scientists are always keen to break a record, and now Professor Xiao Cheng Zeng of the University of Nebraska, Lincoln has theorized a form that would have a density of just 0.6 grams per centimeter cubed, 25 percent lighter than ice XVI.
"We performed a lot of calculations (focused on) whether this is not just a low-density ice, but perhaps the lowest-density ice to date," said Zeng in a statement. "A lot of people are interested in predicting a new ice structure beyond the state of the art."
Zeng is the inventor of “Nebraska Ice”, a form that contracts when frozen, rather than expanding. Icebergs made of Nebraska Ice would sink to the bottom of the ocean, rather than float, which would have been good news for the Titanic, but might have unfortunate effects on ocean ecosystems.
As with a number of other theoretical ice structures, no one has created the structure Zeng has proposed, but in Science Advances he outlines the conditions under which it might occur. The key to Zeng's brainchild is for water to freeze under what is referred to as negative pressure. Instead of normal atmospheric pressure pushing in on the freezing material, the pressure goes outward. Improbable as this sounds, it is not impossible to achieve, but the paper noted, “In the laboratory, applying and maintaining very large tension or negative pressure up to -6000 bar would be very difficult.” Atmospheric pressure is 1.01 bar.
Moreover, the colder the conditions under which the ice forms, the higher the negative pressure required. At 250K (-10°F) the outward pressure would need to be -3411 bar, four times as much as at the bottom of the Mariana Trench. At colder temperatures the pressure required gets even higher, -5834 bar at near absolute zero.
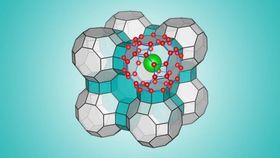
Zeng’s new ice is so light because the water molecules form a near hollow, cage-like structure of 48 molecules.
"Water and ice are forever interesting because they have such relevance to human beings and life," Zeng said. "If you think about it, the low density of natural ice protects the water below it; if it were denser, water would freeze from the bottom up, and no living species could survive. So Mother Nature's combination is just so perfect."