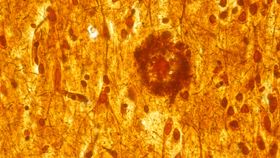
For decades, scientists have identified clumps of sticky proteins that collect in the brains of patients with Alzheimer's disease. These clumps may explain why neurons are lost – and with them, the person’s memory – but the exact mechanism behind how these proteins aggregate have remained elusive.
The rest of this article is behind a paywall. Please sign in or subscribe to access the full content.In a study published in the Journal of Biochemistry, researchers have now traced aggregations of one such protein, ‘tau’, back to a mutation in a common enzyme by mutating fly models. The research is part of an ongoing effort to understand how Alzheimer’s disease develops, and may contribute to a range of other conditions that also result in the buildup of proteins.
Alzheimer’s disease is a debilitating form of dementia that affects more than 5 million Americans. Symptoms of Alzheimer’s include confusion, trouble completing daily tasks, and memory loss. It is a terminal illness that gets progressively worse.
The main theory of Alzheimer’s disease progression suggests large deposits of insoluble proteins either form "plaques" made of a peptide called ?-amyloid or "tangles" made of proteins called tau. Both plaques and tangles usually accompany the development of Alzheimer’s disease, so understanding their formation may be key to unlocking the mystery of how Alzheimer’s disease develops.

In the pursuit of understanding the aggregation of tau, researchers from Tokyo Metropolitan University have identified a key mutation in a gene that may alter the structure of tau, leaving it insoluble and more likely to aggregate. This enzyme, called microtubule affinity-regulating kinase 4 (MARK4), facilitates the detachment of proteins from microtubules (polymers that provide the shape and structure of the cell), which in turn controls cell division, cell cycle control, and – importantly – cell shape alterations. In healthy cells, MARK4 is hard at work detaching microtubules from the cell to allow it to divide and change.
To observe how mutations within the gene change the structure of tau, researchers used drosophila models and mutated MARK4. They then studied the solubility of tau within the brain. In the mutated tau, they found that the tau functioned abnormally and created clumps of insoluble proteins within the brain. With these aberrant clumps of misfolded proteins present, neurons degenerate and die off, which potentially results in the presentation of Alzheimer’s disease.
This research provides a possible mechanism behind tau aggregation in flies, but it remains to be seen whether similar pathways exist in humans. Furthermore, the misfolding of both tau and ?-amyloid is a complex process, so further research will be required to understand just how important this specific mutation is (if at all) in the pathogenesis of Alzheimer’s disease.