Scientists have imaged the coronavirus spike protein in incredible detail, creating the most realistic depiction of the protein in its natural state yet. The study, which was published in Quarterly Reviews Biophysics Discovery, allows scientists to closely view the complete structure of the protein that grants coronaviruses entry into human cells.
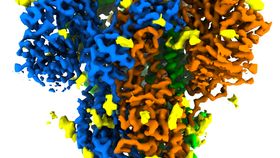
The rest of this article is behind a paywall. Please sign in or subscribe to access the full content.The spike protein, or S protein, is one of the most important functional structures on a SARS-CoV-2 virus particle. If you have ever seen an illustration of a virus, it will likely look like a sphere that is covered with bits that stick out that look like spikes protruding from the surface – this is where the spike protein gets its name. It is this protein that is involved with attaching to the surface of human cells and initiating infection. Specifically, the spike is a glycoprotein (a protein studded with sugar molecules) that binds to the ACE2 receptor on the surface of human cells, granting them entry to reproduce and cause havoc within the body. As seen below in the GIF, the spike is made of 3 protein molecules, each depicted in a different color.

Much like the spike protein is coronavirus’ key to infection, understanding the spike structure is the key to defeating it. All the vaccines against SARS-CoV-2, including the Pfizer/BioTech and Moderna vaccines, provide our bodies with the ‘blueprints’ of the spike protein, which enables the immune system to find a way to defeat it before infection even occurs – check out the vaccine progress with our COVID-19 vaccine tracker for more information.
With the spike being so important in defeating coronaviruses, accurately mapping its structure is crucial. However, much like many proteins, imaging the structure in high resolution poses a difficult challenge – proteins are folded to create extremely complex shapes and most imaging methods require the protein to be taken from its natural state and manipulated prior to visualizing.
In this study, the scientists turned to a more modern microscopy technique called cryogenic electron microscopy (Cryo-EM). Cryo-EM has taken the structural biology field by storm, with the ability to rapidly image proteins to the atomic level in their natural state. By freezing the protein sample to cryogenic temperatures (from -180 degrees Celsius to -269 degrees Celsius), electrons can barrage the sample and provide researchers with a detailed view of the protein. This particular image has an impressive resolution of 3.4Å, which is about the same size as the nucleotides within our DNA.
“The advantage of doing it this way is that when you purify a spike protein and study it in isolation, you lose important biological context: How does it look in an intact virus particle? It could possibly have a different structure there,” said Wah Chiu, a professor at DOE’s SLAC National Accelerator Laboratory and Stanford University and senior author of the study, in a statement.
It is important to note that the spike protein imaged in the study is not of SARS-CoV-2, but instead a more mild relative (HCoV-NL63) that still infects human cells in a similar way.
The image has since been used by the researchers to map 3 new sites where sugar molecules are attached to the spike protein, called glycosylation, which are important in the viruses’ ability to evade the immune system. They now hope to further utilize the technology to study the spike protein on the virus behind COVID-19, although this requires extreme safety and containment procedures.