Along with DNA and RNA, protein is one of the three major biological macromolecules that are essential for all known forms of life. In a remarkable achievement, scientists have now obtained the first-ever photographs of single proteins. As reported in arXiv, this was all possible through the use of a graphene plate.
The rest of this article is behind a paywall. Please sign in or subscribe to access the full content.One of the key aspects of these molecules is that genetic information almost always flows from DNA to RNA to proteins. It may perhaps seem strange, therefore, that something so vital to the existence of life has never been photographed individually before. After all, understanding the structures of proteins is vital for research looking at diseases that involve the incorrect copying or folding of proteins, such as Parkinson’s or Alzheimer’s.
Viewing proteins directly under the microscope is also often problematic, as during the procedure, the molecules are often damaged or destroyed. Imaging techniques that use highly energetic radiation, such as X-ray crystallography, or those using extremely low temperatures, such as cryo-electron microscopy, inevitably alter the structure of the protein.
Focusing high-energy beams onto the proteins will destroy them, so the beams need to be spread out over a larger area. Thus, these two particular methods only give “average” readings of the protein structures, essentially producing blurry images. The proteins themselves are actually quite difficult to keep in one place using traditional glass slides, so imaging them is, for a variety of reasons, tricky.

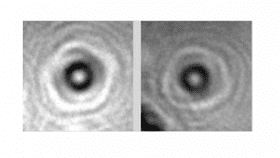
Images of the cytochrome c proteins photographed using the new technique (A). Reconstructions of three separate orientations of the protein using the technique (B) are compared to CGI models of these orientations (C). Jean-Nicolas Longchamp et al./arXiv
This new team of researchers, led by Jean-Nicolas Longchamp of the University of Zurich, Switzerland, has created a new technique that manages to hold these proteins in place without damaging them. First off, low-energy electron beams were used, which means that the proteins won’t be damaged. This, however, means that the reflected beams won’t be able to penetrate through the microscope's detector, but this is where graphene, the proverbial wonder material of the decade, comes in.
“In optical microscopy you have a glass slide. For our electron microscopy we had to find a substrate thin enough to have the electrons passing through,” said Longchamp, as reported by New Scientist. A slide made of graphene, 200 times stronger than steel but only one atomic layer thick, was the perfect candidate.
Using a “holography electron microscope,” the team tested their method on a range of protein samples, all just a few nanometers in size, about ten times smaller than the width of a human hair. Hemoglobin, the protein that transports oxygen in red blood cells, and cytochrome c, the protein that transfers electrons within the body, were just two examples.
The protein remained undamaged and stayed in one place, allowing the researchers to generate images of individual molecules. These “photographs” look incredibly similar to protein images generated from X-ray crystallography, suggesting that these new images are accurate representations of proteins.
This technique provides an excellent stepping stone for medical researchers hoping to understand how individual proteins behave, particularly when they misfold and lead to highly-debilitating diseases.
[H/T: New Scientist]