Cervical cancers are 87 lower among women who were part of the first mass vaccination program in the UK against the human papillomavirus (HPV), compared to previous generations at the same age. The finding, announced in The Lancet, was expected in the light of evidence vaccines control the cancer's predecessors. Nevertheless, with thousands of lives on the line, it's important to be sure, and the results are at the upper end of expectations. The announcement also confirms how much damage was done by anti-vaccination campaigns that hindered programs in some countries.
The rest of this article is behind a paywall. Please sign in or subscribe to access the full content.The overwhelming majority of cases of cervical cancer are caused by HPV infection, mostly by two of the hundreds of strains, which also account for a substantial proportion of rarer cancers in the throat, rectum, and vagina. Consequently, the invention of virus-like particles that stimulate immune responses to the virus was hailed as having the potential to save a quarter of a million lives a year.
However, there is usually a long delay between HPV infection and the development of cancer, and an even longer one from vaccination. This has prevented medical researchers from knowing for certain that the program was working – inevitably seized upon by mass vaccination opponents.
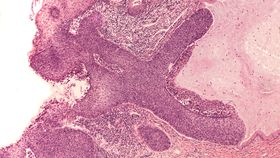
This paper may end such doubts, as much as a single observational scientific study can. The authors used diagnoses for cervical cancer and grade 3 cervical intraepithelial neoplasias (CIN3), which frequently become cancerous between 2006 and 2019 for UK resident women aged 20-64. They compared those who received the vaccine in the original batch at 12-13, and slightly older cohorts who received catch-up vaccinations at 14-18, to those who were adults before the vaccine became available.
Using a data pool of more than 13 million years of follow-up, those who got the vaccine at 12-13 had an 87 percent reduction in cancers compared to those who missed out on vaccination entirely at equivalent ages. The intermediary groups had reductions of 62 and 34 percent depending on the age at which they were vaccinated. As a result, the authors calculate, 448 cervical cancers and more than 17,000 CIN3s, were prevented in the UK through vaccination by June 30, 2019, and the numbers will grow rapidly as more women reach the ages where cervical cancer was previously most common.
“It’s been incredible to see the impact of HPV vaccination and now we can prove it prevented hundreds of women from developing cancer in England,” lead author Professor Peter Sasieni of Kings College London said in a statement. “We’ve known for many years that HPV vaccination is very effective in preventing particular strains of the virus, but to see the real-life impact of the vaccine has been truly rewarding."
The findings refute the argument made by some fence-sitters that HPV vaccination should occur, but later. HPV is transmitted sexually and critics alleged, in the words of Australia's Deputy Prime Minister, the vaccine will give 12 year-olds “a license to be promiscuous”. The choice of year eight was made on the basis that vaccinations were necessary before any members of a class became sexually active, even if most would not start until many years later.
Numerous peer-reviewed studies have provided evidence HPV vaccination was likely to cut cervical cancers, starting with those that showed reduced infections, sharp reductions in genital warts, and finally pre-cancerous cells. Most of these were conducted with the Gardasil vaccine, which was more expensive but targeted four HPV strains (more recently increased to nine) rather than Cevarix's two.