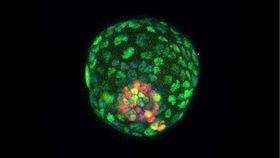
A groundbreaking study has seen scientists create the early stages of artificial mice embryos in a lab using stem cells, rather than sperm or eggs, for the first time.
The rest of this article is behind a paywall. Please sign in or subscribe to access the full content.It’s still very early days for the research, and it has some big problems to iron out, but it could potentially pave the way towards creating viable embryos just from cultured cells. It could also provide scientists with an indefinite source of blastocysts to study the early stages of development, without the need for sperm, eggs, or natural embryos.
“These studies will help us to better understand the very beginnings of life; how early on in life a single cell can give rise to millions of cells and how they are assembled in space and time to give rise to a fully developed organism. Importantly, this work avoids the use of natural embryos and is scalable,” Juan Carlos Izpisua Belmonte, study author and professor in Salk’s Gene Expression Laboratory, explained in a statement.
Reporting in the journal Cell, researchers at the Salk Institute and the University of Texas Southwestern Medical Center created structures resembling mouse blastocysts, known as blastoids.
Blastocysts are structures made up of 100+ cells or so, found in the very early stages of development in mammals. After the blastocyst implants itself into the uterus, it can develop into an embryo. Typically, the blastocyst forms around five days after a sperm has fertilized an egg.
However, the researchers managed to create an early form of blastocyst using pluripotent stem cells, “master cells” that can turn into almost any kind of tissue in the body, obtained from adult mouse cells. The stem cells were placed together and nurtured in a special culture medium. They soon formed connections with each other, eventually forming a ball of cells with an inner and outer layer.
The structure of this, according to the researchers, was remarkably like a natural blastocyst. The artificial blastoids contained the same three primordial cell types found in natural blastocysts, along with being a similar size and similar gene expression.
Other researchers working in the field have described the research as “wonderful,” “significant,” and “outstanding,” although they warn there are still many hurdles to overcome before we see this research being applied.
For example, it’s noteworthy that the blastocyst-like structures showed signs of malformation and disorganization, meaning they wouldn’t be able to develop into a fully viable embryo or mouse.
“I would caution against interpreting the current study as showing that embryos can be made from adult tissues – this has the potential to raise undue ethical concerns, the structures made are not embryos, and the research is in mice not humans,” said Dr Harry Leitch, a stem cell biologist at the MRC London Institute of Medical Sciences and Imperial College London who was not involved in the study.
“In reality, this study reports incremental findings in a pre-existing model system, and makes a valuable contribution to ongoing research in the field.”