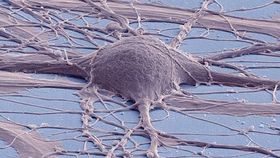
A study published in Cell Stem Cell has demonstrated that implanted neural stem cells can form connections not only between themselves but also with the host’s own neural network in mice.
The rest of this article is behind a paywall. Please sign in or subscribe to access the full content.Repairing spinal cord injuries has proven to be one of the greatest challenges in modern medicine. Improvements have been made in recent years, with attempts including an implant that "reads" a patient's brain signals and translates them to movement, but perhaps the most promising has been stem cell grafts.
Researchers from the University of California San Diego School of Medicine used neural stem cells to "patch" up the damaged spinal cord of mice. They then used a specialized technique called calcium imaging to follow the development of new stem cells as they integrated into the host's nervous system. Once the cells were grafted to the injury, the stem cells formed connections between each other that could be activated by signals from the host. Stimuli from the host brain even elicited responses in neurons below the injury site.
The results are the first time researchers have confirmed connectivity between implanted cells and host cells, and may pave the way for stem cell graft therapies in the future. Previous research has only demonstrated that some function could be regained in animals with spinal cord injury, but never confirmed functional connections were being made by implanted stem cells.
"We knew that damaged host axons grew extensively into (injury sites), and that graft neurons in turn extended large numbers of axons into the spinal cord, but we had no idea what kind of activity was actually occurring inside the graft itself," said Steven Ceto, a postdoctoral fellow, in a statement. "We didn't know if host and graft axons were actually making functional connections, or if they just looked like they could be."
This was all made possible by recent advances in calcium imaging techniques, which allowed the researchers to record the signals between nerve cells. Calcium is an integral part of how the brain communicates through neurons, so by tracking the levels of calcium in cells, they could map connections made by grafted stem cells to host neurons. To stimulate and record the neurons, the scientists used a technique called optogenetics, which uses light (rather than electricity) to activate or inhibit specific neuron populations.
Limitations of the study include measurements that were not made. Ceto and colleagues suggest other inputs, not just those from the corticospinal system (the pathway leading from the brain to motor neurons in the spinal cord), may also play a role in shaping how grafts react to stimuli. Although the study is in its early days and only in mice, the researchers hope to push grafting therapy forward in the hope of helping spinal cord injury patients. Their research will focus on enhancing the application of stem cell grafts and encouraging the generation of new connections via electrical stimulation.
"While the perfect combination of stem cells, stimulation, rehabilitation and other interventions may be years off, patients are living with spinal cord injury right now," said Mark H. Tuszynski, professor of neurosciences and director of the Translational Neuroscience Institute at UCSD School of Medicine. "Therefore, we are currently working with regulatory authorities to move our stem cell graft approach into clinical trials as soon as possible. If everything goes well, we could have a therapy within the decade."