An international consortium of researchers—led by a group from the University of Leicester—have identified key steps in cell division that could reveal new targets for cancer therapy. The two papers, published in the Journal of Cell Biology, provide new insights into the mechanics of cell division, how it goes wrong in cancer and how scientists could tailor drugs to target the process, which may effectively eradicate the tumor.
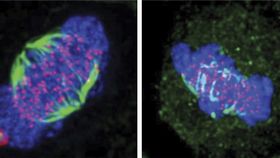
The rest of this article is behind a paywall. Please sign in or subscribe to access the full content.Cell division, the process where a parent cell divides into two or more daughter cells, requires the assembly of a rugby ball-shaped scaffold, known as the mitotic spindle. The spindle is composed of fibers called microtubules that originate from two distinct structures called centrosomes. These fibers capture the duplicated chromosomes and separate them apart. Researchers have found out how the enzyme Nek6 controls the stability of the structural scaffold and contributes to the separation of centrosomes.
The first of these two papers, led by Dr. Laura O'Regan, shows that Nek6 modifies a chaperone, which are proteins that act as "cellular guardians," folding proteins into their correct shape and assembling them into functional complexes. The chaperone, known as Hsp72, is loaded onto spindle microtubules, which are essential for robust spindle integrity.
“We show that in the absence of Nek6 or Hsp72, fragile and misshapen spindles are formed that cannot pull the chromosomes apart and so cells get stuck in the process of cell division,” says Professor Andrew Fry, director of research in the College of Medicine, Biological Sciences and Psychology at Leicester, who led the research.
There is a growing interest in these chaperones by researchers and the pharmaceutical industry as they also protect cancer cells against a stressful environment and keep them alive.
“Cancer cells frequently have more than the normal number of chromosomes, a feature that not only makes them behave abnormally but which also causes cell division to be more difficult to execute. As a result, they often increase the amount of the proteins required for cell division, such as Nek6 and Hsp72,” Fry tells IFLScience.
The second paper, led by Dr. Suzanna Prosser, shows that the enzyme Nek5, which belongs to the same family as Nek6, plays a key role in making sure that centrosomes separate at the right time. In the absence of their function, Fry says, centrosomes separate late, leading to “errors in the division of genetic material that can promote cancer.”
These proteins could provide researchers with an opportunity to kill the tumors by targeting them with drugs that inhibit these proteins. Fry suggests that a new generation of targeted drugs that interfere with these particular proteins may offer new avenues for cancer therapy.
“We will engage with academic and industrial partners to develop specific inhibitors of these enzymes that can be used to test our hypothesis that inhibiting these activities can lead to selective killing of cancers in different model systems," says Fry. "If these proof-of-principle studies prove promising, then we would expect to launch the first clinical trials for drugs against these targets in cancer patients."