A treatment that was awarded Breakthrough Therapy designation by the FDA last year is once again raising hopes for those with B cell cancers. In a recently completed trial, eight out of 14 leukemia patients responded to the novel treatment, a type of personalized cell therapy. Encouragingly, some also went into remission and have remained cancer-free for more than four and a half years.
The rest of this article is behind a paywall. Please sign in or subscribe to access the full content.The trial actually began five years ago and is one of a small handful of trials designed to test the University of Pennsylvania's novel immunotherapy, called CTL019, on patients with cancers of the immune system called B cell cancers. In this particular trial, participants had chronic lymphocytic leukemia (CLL) that had either relapsed or failed to respond to numerous conventional treatments.
CLL affects white blood cells called lymphocytes, causing under-developed cells to proliferate uncontrollably in the body. Because these cells aren’t mature, they are unable to function correctly and thus leave the patient vulnerable to infection. Furthermore, the overproduction of these cells comes with another cost: a reduction in the generation of other blood cells, like red blood cells, which can cause anemia or bleeding problems.
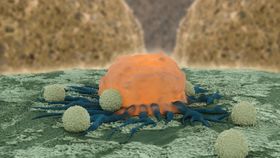
Although the immune system is designed to detect and eliminate invaders and diseased cells, unfortunately cancerous cells are often able to slip under its radar. This is where CTL019 comes in. This is actually a personalized therapy in which the patient’s own immune cells are removed and reprogrammed into cancer cell-stalkers.
More specifically, they extract a type of lymphocyte called a T lymphocyte and engineer them to produce an antibody-like protein called a chimeric antigen receptor (CAR). This recognizes and binds to a protein called CD19 which is found on the surface B cells, including those of several different leukemias and lymphomas, ultimately resulting in cell death.
After tweaking the cells, the patient’s white blood cells are depleted by drugs and replaced by this modified batch. And encouragingly, it seems to be an effective approach in some patients.
As described in Science Translational Medicine, four of the 14 patients in the pilot trial achieved remission, one of whom has now been cancer-free for five years. While one patient unfortunately died during remission as a result of an infection following tumor-removal surgery, the other two have been in remission for more than two years despite receiving no further treatments.
Alongside these patients, a further four also responded to the therapy, although only partially. Three of these patients ultimately died, although one is still alive three years later after receiving other treatments.
The final six patients did not respond to the treatment, with some experiencing cancer progression just one month following infusion of the cells. In these patients, the reprogrammed T cells were found to not establish themselves sufficiently in the immune system.
While it’s easy to dwell on the negatives, this was only a small trial, and achieving remission for years after treatment is a significant result. Furthermore, other trials involving different cancers have also witnessed positive results. For example, more than 90% of pediatric patients with acute lymphoblastic leukemia responded to the treatment, and 100% of adults with follicular lymphoma.
Cancer is not a single disease, so there will be no one miracle cure. But we can look upon these results as an encouraging step in the right direction for B cell cancers.