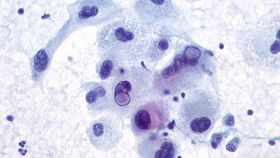
A genetically engineered herpes virus can both kill aggressive cancer cells and kick-start an immune response against them, scientists are reporting. Following the world's first investigation of viral therapy to treat skin cancer, the novel treatment was found to increase the survival of some patients by several years. The study, published in the Journal of Clinical Oncology, is the largest ever randomized trial of viral anti-cancer therapy.
The rest of this article is behind a paywall. Please sign in or subscribe to access the full content.The Institute of Cancer Research (ICR) in the U.K. led an international consortium of researchers to trial a modified version of a herpes virus, called Talimogene Laherparepvec (T-VEC), on 436 patients who had inoperable malignant melanoma. One group of patients were injected with T-VEC, while the others were given a control immunotherapy, a type of treatment that uses the body's immune system to fight disease.
“We may normally think of viruses as the enemies of mankind, but it’s their very ability to specifically infect and kill human cells that can make them such promising cancer treatments,” said Professor Paul Workman, chief executive of the ICR, in a statement.
"In this case we are harnessing the ability of an engineered virus to kill cancer cells and stimulate an immune response. It’s exciting to see the potential of viral treatment realized in a Phase III trial, and there is hope that therapies like this could be even more effective when combined with targeted cancer drugs to achieve long term control and cure,” he added.
Researchers modified the virus by removing two key genes, called ICP34.5 and ICP47, to make it harmless to healthy cells. The virus can, however, still replicate within cancer cells and destroy them by bursting them open. T-VEC has also been genetically engineered to release a molecule called GM-CSF, which prompts a rigorous response from the immune system.
The results showed that 16.3% of the group given T-VEC responded to treatment and were still in remission after six months. In comparison, 2.1% of the control group showed the same response. The 163 patients with stage 3 and early stage 4 melanoma who were given T-VEC had an average survival of 41 months, while 66 earlier-stage patients in the control group lived for 21.5 months. Some patients in the viral therapy group had a response extending past three years—a benchmark used by oncologists to define an effective treatment.
“There is increasing excitement over the use of viral treatments like T-VEC for cancer, because they can launch a two-pronged attack on tumors – both killing cancer cells directly and marshaling the immune system against them. And because viral treatment can target cancer cells specifically, it tends to have fewer side-effects than traditional chemotherapy or some of the other new immunotherapies,” said U.K. trial leader Kevin Harrington, professor of Biological Cancer Therapies at the ICR, in a statement.
The study found that patients with less advanced cancers had a more significant response to the viral therapy, which Harrington suggests is "encouraging" as T-VEC could play an important role as a first-line of defense against more aggressive cancer. T-VEC has been submitted to both the U.S. Food and Drug Administration and the European Medicines Agency. If approved, researcher hope similar viral treatments could be used to tackle other cancers.
Image credit: Malignant Melanoma by Ed Uthman, via Flickr. CC BY 2.0