Need some new muscle? We’ll print that for you.
The rest of this article is behind a paywall. Please sign in or subscribe to access the full content.In a tremendous stride forwards for the field of regenerative medicine, researchers in the U.S. have managed to invent a 3D printer capable of churning out human-scale, living tissues that survived and integrated when implanted into an animal. While it’s still early days, the team demonstrated that the tissues developed the right strength and function required for human application, raising the possibility that the technique could one day be used to help people whose tissue has been damaged by disease or trauma.
The research is published in the journal Nature Biotechnology.
“We know from other industries that 3D printers can print objects of any size and shape,” senior author Anthony Atala from the Wake Forest Institute for Regenerative Medicine told IFLScience. “When printing human tissues and organs, of course, we need to make sure the cells survive, and function is the final test. Our research indicates the feasibility of printing bone, muscle, and cartilage for patients. We will be using similar strategies to print solid organs.”
Researchers are increasingly looking towards 3D printing in regenerative medicine because of the ease and precision it offers for creating an array of complex structures that can be tailored to the patient. While it’s already possible to 3D print solid objects that can be used to replace bone, with one woman even receiving a plastic skull, creating living tissues represents a much greater challenge. Unlike real organs, a lack of blood vessels to keep the inner cells happy imposes a size limit that essentially means the tissues can never be made big enough to be clinically relevant.
On top of that, problems with structural integrity mean that the printed tissues are often too flimsy to survive after being implanted. But the group from the Wake Forest Baptist Medical Center might have come up with a way to overcome these issues. Describing their developments in the paper, the researchers bestowed their tissues with mechanical stability by printing patterns of live cells in a mix that contained biodegradable, plastic-like materials. These were designed to dissolve in the body after the construct has had enough time to grow and mature into a structure capable of retaining its shape.

The 3D printer hard at work. Credit: Wake Forest Institute for Regenerative Medicine
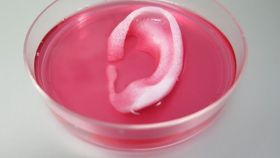
To help keep the cells alive, the team printed a network of tiny channels throughout the structure which allowed the flow of oxygen and nutrients until blood vessels can work their way in post-implantation and offer their own supply. This was demonstrated after they inserted a human-sized ear, made by printing cartilage cells into a design dictated by a CT scan, under the skin of mice. Two months after the surgery, the structures had maintained their shape, matured into cartilage tissue, and showed signs of vascularization.
In another proof-of-concept experiment, the team printed rat muscle cells into supportive structures and left them to grow for a week before implanting them into rats. Amazingly, after just two weeks the construct had matured and developed organized muscle fibers which made contacts with nerves, an essential feature if the tissue is going to gain any functionality. Indeed, electrically stimulating the muscle caused it to respond and fire up neighboring neurons to roughly the same extent that would be expected for immature muscle.
Longer-term studies are needed to investigate the lifespan of these structures, but the work is encouraging nonetheless, and as a patient’s own cells could be used, the common issue of rejection would be negated.