The ultimate goal of stem cell research is to create functional replica tissues and organs for use as replacements in times of injury or disease, or for use in the development of drugs and other therapeutic techniques. Getting tissues to grow in the lab in three dimensions has been challenging across the board, but this is especially problematic for structures in the nervous system. Beyond getting the neurons to grow at all, they must be connected in a very particular manner in order to function. A major step forward has been taken on this front by a team from RIKEN Center for Developmental Biology in Japan, who state in Cell Reports that they have successfully grown 3D functional brain tissue that has even grown with proper patterning.
The rest of this article is behind a paywall. Please sign in or subscribe to access the full content.The brain tissues were grown from human embryonic stem cells, and growth factors were added in series throughout development. The first was basic fibroblast growth factor 2 (FGF2), which plays a variety of roles in development, wound healing, and even tumor growth. Over a course of three weeks, the cells began to differentiate into midbrain and hindbrain regions. In the two weeks that followed, the epithelial cells that give rise to the cerebellum appeared in the hindbrain. Those cells also exhibited precursors to specialized neurons exclusive to the cerebellum, like Purkinje cells that help regulate motor movement and granule cells, which have a wide range of functions.
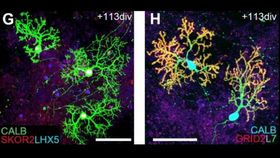
The researchers also added FGF19 in the second week and found that it led to indications of dorsal-ventral (top-bottom) patterning within 21 days. During the 4th week of culture, stromal cell-derived factor 1 (SDF1) was added, creating the proper dorsal-ventral polarity in the growing brain structure. In the dorsal region, SDF1 led to the development of areas that would produce Purkinje, deep cerebellar projection neurons and the rhombic lip, which is essential for the development and migration of granule cells.
After 15 weeks of culturing, the cells treated with FGF2 showed additional markers for Purkinje cells, in addition to beginning to take on their physical characteristics and rudimentary electrophysiological function. The granule cells also appeared more mature, and there was even evidence of normal cell migration. The developing neuronal cells were even growing in the proper orientation to one another.
The self-patterning of cells seen in this study was similar to what can be expected during the first trimester of embryonic development, though the recreation is not complete. They hope to refine this in the future, improving what can be achieved in the span of the first trimester. Ultimately, they hope to devise a method which will allow for long-term culture of the brain tissue through the end of the second trimester as well.
“The principles of self-organization that we have demonstrated here are important for the future of developmental biology,” lead author Keiko Muguruma said in a press release. "Attempts to generate the cerebellum from human iPS [induced pluripotent stem] cells have already met with some success, and these patient-derived cerebellar neurons and tissues will be useful for modeling cerebellar diseases such as spinocerebellar ataxia.”
The study’s senior author, Yoshiki Sasai, committed suicide last August, following the highly-publicized retraction of two related papers he co-authored that were originally published in Nature in January 2014 after a Japanese government panel deemed the studies contained falsified data. The researchers vehemently denied intentional wrongdoing.